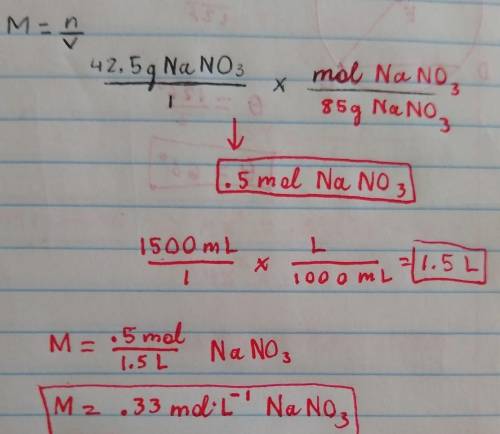Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:00, haydjanggg6578
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2


Chemistry, 23.06.2019 07:30, lifeislove3251
Type the letter that represents the correct location for each particle type below. the neutron is found at __ the electron is found at __ the proton is found at __
Answers: 1
Do you know the correct answer?
A given solution has 42.5 g NaNO3 in 1500 mL of water. What is the molarity of this solution? Show y...
Questions in other subjects:

Mathematics, 28.04.2021 18:20




English, 28.04.2021 18:20



Business, 28.04.2021 18:20

Mathematics, 28.04.2021 18:20

Mathematics, 28.04.2021 18:20