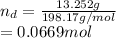Chemistry, 22.07.2021 22:40, lukecoupland4401
An intravenous solution was prepared by add-in 13.252 g of dextrose (C6H12O6) and 0.686 g of sodium chloride to a 250.0 mL volumetric flask and diluting to the calibration mark with water. What is the molarity of each component of the solution

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, claudia122752
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1



Chemistry, 23.06.2019 00:30, hdhshshs741
An unknown insoluble substance displaced the water shown. it's mass is indicated on the triple beam balance. mass = a. 694 b. 693.5 c. 693.0 d.693.8
Answers: 1
Do you know the correct answer?
An intravenous solution was prepared by add-in 13.252 g of dextrose (C6H12O6) and 0.686 g of sodium...
Questions in other subjects:




English, 05.05.2020 10:11



Computers and Technology, 05.05.2020 10:11





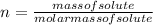
 ) is:
) is: