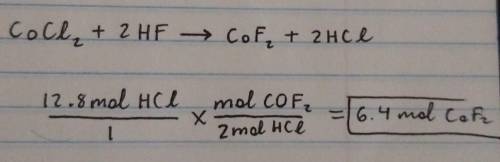Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 18:00, jalenclarke25
What volume would 2.25 moles of ne has occupy at stp?
Answers: 1

Chemistry, 22.06.2019 18:30, chinadoll24
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2

Chemistry, 22.06.2019 21:00, rah45
Which of these is an example of pseudoscience? a) predicting the time of sunrise based on data on position of earth b) predicting the date of the moon phases based on data on position of earth c) predicting eclipses based on the position of the sun and the moon d) predicting future events in a person's life based on the position of the moon
Answers: 1
Do you know the correct answer?
Calculate the amount of mole(s) of CoF2 required to react with 12.8 moles of HCI.
CoCl2 + 2HF -->...
Questions in other subjects:


Mathematics, 20.12.2020 20:20

Social Studies, 20.12.2020 20:20



Business, 20.12.2020 20:20

English, 20.12.2020 20:20


Health, 20.12.2020 20:20

Chemistry, 20.12.2020 20:20