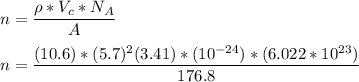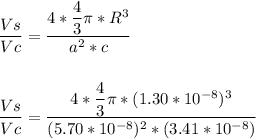Chemistry, 20.07.2021 03:00, corbin3582
Consider a hypothetical metal that has a density of 10.6 g/cm3, an atomic weight of 176.8 g/mol, and an atomic radius of 0.130 nm. Compute the atomic packing factor if the unit cell has tetragonal symmetry, values for the a and c lattice parameters are 0.570 and 0.341, respectively.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, breannaasmith1122
If 1.63 times 10 negative 4 of helium dissolves in 100.0g of water, what is the concentration in parts per million
Answers: 3

Chemistry, 22.06.2019 23:00, autumperry3599
What is the chemical formula for dihydrogen monoxide
Answers: 2

Chemistry, 23.06.2019 06:00, asalimanoucha2v
•what conclusions can you make about the relationship between the volume of a gas and its temperature? • what conclusions can you make about the relationship between the volume of a gas and its pressure? • what possible variables have you not accounted for? as you did the procedures, is it possible that the atmospheric pressure may have changed? if it did change over the course of your experiment, then how would your results have been affected?
Answers: 3

Chemistry, 23.06.2019 07:30, danielahumajova6
How do you interpret a chromagram for what mixtures contain?
Answers: 1
Do you know the correct answer?
Consider a hypothetical metal that has a density of 10.6 g/cm3, an atomic weight of 176.8 g/mol, and...
Questions in other subjects:


SAT, 12.12.2020 16:50


Mathematics, 12.12.2020 16:50

Arts, 12.12.2020 16:50



Chemistry, 12.12.2020 16:50

Mathematics, 12.12.2020 16:50