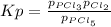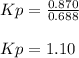A reaction vessel is charged with phosphorus pentachloride, which partially decomposes to phosphorus trichloride and molecular chlorine according to the following reaction:
PCl5(g)â PCl3(g)+Cl2(g)
When the system comes to equilibrium at 250.0°C, the equilibrium partial pressures are: PPCl5 = 0.688 atm and PPCl3 = PCl2 = 0.870 atm.
Required:
What is the value of Kp at this temperature?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:50, kyleighmarie05
H2so4(aq) + mg(s)—> mgso4(aq) +h2(g) which substance is the acid in the reaction?
Answers: 3


Chemistry, 22.06.2019 13:30, justinerodriguz2878
What are the major types of a chemical compound
Answers: 2

Chemistry, 22.06.2019 15:00, alanmarcus22
What does the symbol (–hfus) indicate in a phase change?
Answers: 1
Do you know the correct answer?
A reaction vessel is charged with phosphorus pentachloride, which partially decomposes to phosphorus...
Questions in other subjects:



Social Studies, 03.07.2019 17:30

Mathematics, 03.07.2019 17:30

Mathematics, 03.07.2019 17:30

Mathematics, 03.07.2019 17:30

Biology, 03.07.2019 17:30

English, 03.07.2019 17:30


English, 03.07.2019 17:30