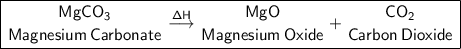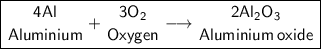Chemistry, 16.07.2021 06:00, rorathan123
Complete and balance the following chemical equations. Identify the reaction type as: combination, decomposition, single replacement, double replacement, or combustion.
Products:
Magnesium Oxide + Carbon dioxide.
a) MgCO₃ (Heat is supplied to the reaction (triangle over a arrow) -> Reaction type:
Products:
Aluminum Oxide
b) Al + O₂ -> Reaction type:

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 01:30, bgarrison364
What is produced from neutralization of an acid and a base? a. hydronium ions b. citric acid c. salt and water
Answers: 1
Do you know the correct answer?
Complete and balance the following chemical equations. Identify the reaction type as: combination, d...
Questions in other subjects:



Mathematics, 15.01.2021 08:30

Social Studies, 15.01.2021 08:30




Advanced Placement (AP), 15.01.2021 08:30


English, 15.01.2021 08:30