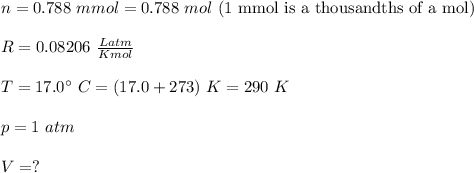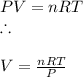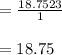Chemistry, 15.07.2021 21:00, bellapimienta8
A reaction at evolves of dinitrogen monoxide gas. Calculate the volume of dinitrogen monoxide gas that is collected. You can assume the pressure in the room is exactly . Be sure your answer has the correct number of significant digits.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, ashlynneboogs0056
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 23.06.2019 01:30, rubyr9975
What is the importance of interlocking the fingers and rubbing while washing hands? the palms are the dirtiest parts of the hands. the spaces between the fingers get washed. the backs of the hands get washed. the fingernails are the dirtiest parts of the hands
Answers: 1

Chemistry, 23.06.2019 04:00, ayoismeisjjjjuan
How many liters of water can be produced from 5.0liters of butane gas at stp, assuming excess oxygen? c4h10(g) + 02(g) → co2 (e) + h2o (g)
Answers: 2
Do you know the correct answer?
A reaction at evolves of dinitrogen monoxide gas. Calculate the volume of dinitrogen monoxide gas th...
Questions in other subjects:

Mathematics, 18.06.2021 15:50

Mathematics, 18.06.2021 15:50

Computers and Technology, 18.06.2021 15:50

Mathematics, 18.06.2021 15:50


Mathematics, 18.06.2021 15:50

History, 18.06.2021 15:50




 ".
".