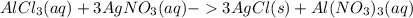Chemistry, 12.07.2021 19:30, swaggyd6603
If 13.50 mL of an aluminum chloride solution is needed to reach the equivalence point with 10.00 mL of 0.109 M silver nitrate solution, determine the molarity of the aluminum chloride solution.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, cynthiagutierrez65
Given that the molar mass of nano3 is 85.00 g/mol, what mass of nano3 is needed to make 4.50 l of a 1.50 m nano3solution? use .6.75 g18.9 g255 g574 g
Answers: 1



Do you know the correct answer?
If 13.50 mL of an aluminum chloride solution is needed to reach the equivalence point with 10.00 mL...
Questions in other subjects:



History, 18.08.2019 12:30

Mathematics, 18.08.2019 12:30

Mathematics, 18.08.2019 12:30

Geography, 18.08.2019 12:30

Physics, 18.08.2019 12:30


Mathematics, 18.08.2019 12:30

Mathematics, 18.08.2019 12:30