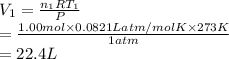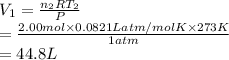Chemistry, 12.07.2021 05:40, mirzakasumovic8926
Consider an experimental run at 273 K where the initial number of moles (n1) is actually 1.00 mol, and the final number of moles (n2) is 2.00 mol. Use the simulation to find the volume (V1) of 1.00 mol of helium at 273 K, and calculate the final volume (V2). Express the volume to three significant figures, and include the appropriate units.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, awdadaddda
How air particles exert a pressure on the inside of the balloon
Answers: 1


Chemistry, 22.06.2019 07:20, JKINGblackstar3502
After watching the video "zinc strip in copper nitrate solution", and reading the instructions, click on the link labeled "start" just below the drawing of the pencil tip. follow the direction to complete the 3x3 grid. answer the below questions for the portion of the activity in which sn(s) is placed in agno3(aq)
Answers: 1
Do you know the correct answer?
Consider an experimental run at 273 K where the initial number of moles (n1) is actually 1.00 mol, a...
Questions in other subjects:






Health, 20.05.2021 15:50

Mathematics, 20.05.2021 15:50

Mathematics, 20.05.2021 15:50


Mathematics, 20.05.2021 15:50


 of 1.00 mol of helium at 273 K is 22.4 L and the final volume
of 1.00 mol of helium at 273 K is 22.4 L and the final volume 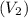 is 44.8 L.
is 44.8 L.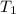 = 273 K,
= 273 K, 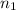 = 1.00 mol
= 1.00 mol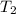 = 273 K,
= 273 K,  = 2.00 mol
= 2.00 mol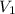 and
and 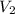 is calculated as follows.
is calculated as follows.