Chemistry, 08.07.2021 21:50, jjiopppotdd5638
For a reaction, AH = 176 kJ/mol and A SO = 0.285 kJ/(K•mol). At what
temperatures is this reaction spontaneous?
A. At no temperature
B. T< 50 K
C. T>617 K
D. T< 617 K

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, badgirl2005
This is a mixture that has the same composition throughout.
Answers: 1


Chemistry, 23.06.2019 02:00, jacckiie5176
Which of these is a density dependent factor? a. epidemic b. earthquake c. drought d. hurricane
Answers: 2

Do you know the correct answer?
For a reaction, AH = 176 kJ/mol and A SO = 0.285 kJ/(K•mol). At what
temperatures is this reaction...
Questions in other subjects:

Mathematics, 07.09.2021 09:20


Chemistry, 07.09.2021 09:20

Mathematics, 07.09.2021 09:30

History, 07.09.2021 09:30

Mathematics, 07.09.2021 09:30


Social Studies, 07.09.2021 09:30


Mathematics, 07.09.2021 09:30


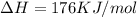
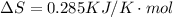
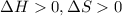
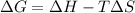
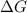 is negative, then the reaction is spontaneous.
is negative, then the reaction is spontaneous.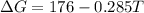
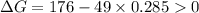
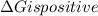 .Hence, the reaction is not spontaneous.
.Hence, the reaction is not spontaneous.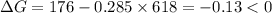
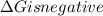 .Hence, the reaction is spontaneous.
.Hence, the reaction is spontaneous.