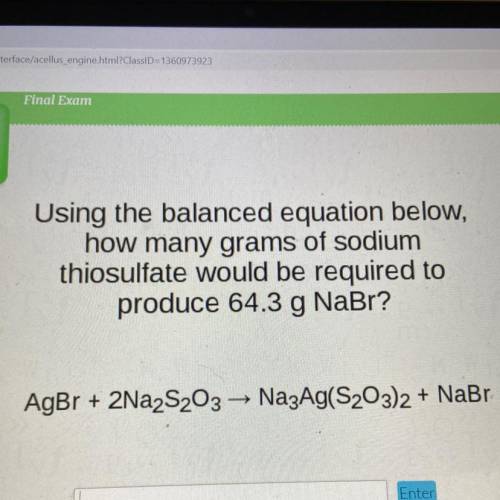
Using the balanced equation below,
how many grams of sodium
thiosulfate would be required to<...

Chemistry, 06.07.2021 19:00, destinyranson
Using the balanced equation below,
how many grams of sodium
thiosulfate would be required to
produce 64.3 g NaBr?
AgBr + 2Na2S203 — Na3Ag(S203)2 + NaBr

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:30, colochaortiz20p7cajw
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 00:00, rileyallen4186pd5tgy
Several kinds of bears are found on earth. most bears are brown or black, but one type of bear, the polar bear, is white. what process led to this difference in fur color? explain your answer.
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Mathematics, 13.03.2020 16:50




Chemistry, 13.03.2020 16:51


English, 13.03.2020 16:51






