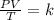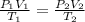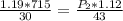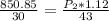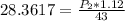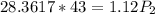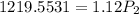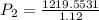Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:00, lindseyklewis1p56uvi
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1


Chemistry, 23.06.2019 04:00, Mitchmorgan3816
Why must humans find substitutes for many minerals found on earth? (a) form at an extremely slow rate (b) controlled by other countries (c) too deep in the earth to collect
Answers: 1
Do you know the correct answer?
calculate the final pressure of a gas that is expanded from 725cm³ at 30C and 1.19 atm to 1.12cm³ at...
Questions in other subjects:



Geography, 22.07.2019 20:30


Chemistry, 22.07.2019 20:30




Mathematics, 22.07.2019 20:30

Mathematics, 22.07.2019 20:30