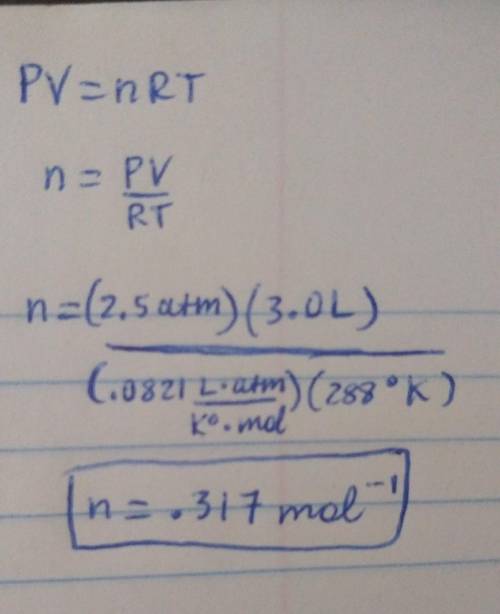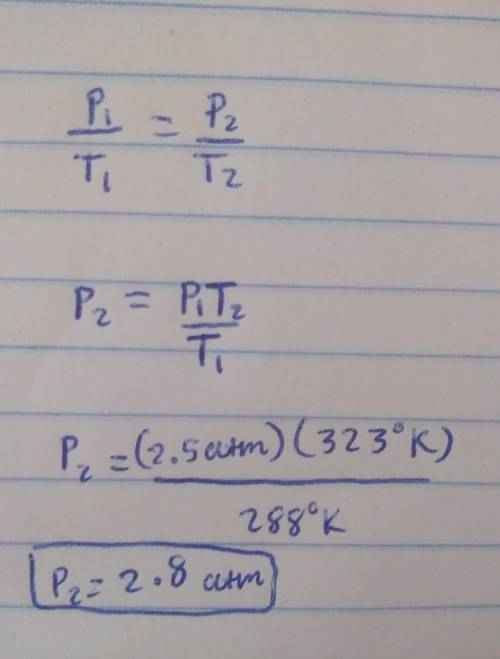Chemistry, 03.07.2021 01:30, emilylunaa
A 3.0-liter sample of an ideal gas is at a pressure of 2.5 atm at 15oC. (i) How many moles of gas are in the sample? (ii) If the volume does not change, what is the pressure of the gas when the temperature is 50oC?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, gatorr2010
An alcohol thermometer makes use of alcohol's changing in order to measure temperature. as the temperature goes up, the alcohol contained in the thermometer increases in volume, filling more of the thermometer's tube.
Answers: 3

Chemistry, 21.06.2019 22:00, pettygirl13
Describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? )
Answers: 3


Chemistry, 22.06.2019 16:00, hjgjlgkjg
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
Do you know the correct answer?
A 3.0-liter sample of an ideal gas is at a pressure of 2.5 atm at 15oC. (i) How many moles of gas ar...
Questions in other subjects:

Mathematics, 08.12.2021 15:30

Mathematics, 08.12.2021 15:30

Mathematics, 08.12.2021 15:30


Law, 08.12.2021 15:30


Computers and Technology, 08.12.2021 15:30

French, 08.12.2021 15:30

English, 08.12.2021 15:30