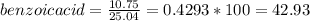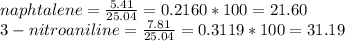Chemistry, 02.07.2021 04:40, lydiapoetz5330
1. Calculate the percent recovery of benzoic acid, naphthalene and 3-nitroaniline if you were able to collect 10.75 g of benzoic acid, 5.41 g of naphthalene, and 7.81 g of 3-nitroaniline from a set of extractions. The starting mass of the mixture was 25.04 g. (0.6 pt) 2. Describe why it is important to use sodium hydroxide and hydrochloride acid in this experiment. Why was it necessary to initially start off with a 5% solution of the acid or base for this experiment

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 12:30, ramireznaidelyn
4nh3+5o2--> 4no+6h20what is the total number of moles of h2o produced when 12 mole of nh3 is completely consumed?
Answers: 3



Chemistry, 22.06.2019 19:30, toriabrocks
If 16.00g of hydrogen gas reacts with 126.73g of oxygen, how many grams of water are yielded? (both reactants are completely consumed in the reaction.)
Answers: 2
Do you know the correct answer?
1. Calculate the percent recovery of benzoic acid, naphthalene and 3-nitroaniline if you were able t...
Questions in other subjects:


History, 06.01.2022 16:10

History, 06.01.2022 16:10

Mathematics, 06.01.2022 16:10

History, 06.01.2022 16:10


Mathematics, 06.01.2022 16:10


Mathematics, 06.01.2022 16:10

Mathematics, 06.01.2022 16:10