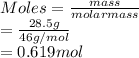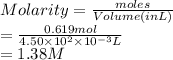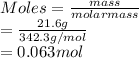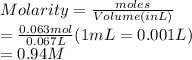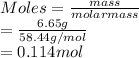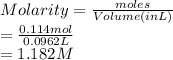Be sure to answer all parts.
Calculate the molarity of each of the following solutions:
(a) 2...

Chemistry, 01.07.2021 05:10, aliceotter2007
Be sure to answer all parts.
Calculate the molarity of each of the following solutions:
(a) 28.5 g of ethanol (C2H5OH) in 4.50 x 102 mL of solution
M M
es
(b) 21.6 g of sucrose (C12H22011) in 67.0 mL of solution
M
(c) 6.65 g of sodium chloride (NaCl) in 96.2 mL of solution
M

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, darg3990rgp2t0r2
Does anyone know a lot about how to: - calculate mass of magnesium metal - calculate the actual yield of magnesium oxide - calculate the theoretical yield of mgo - calculate the percent yield of mgo - determine the percent yield of mgo - determine the average percent yield of mgo i had to do an online lab and its asking these questions but i have no idea where to start or how to be able to find these things. i can post the chart of the data from the lab or if you can tell me exactly how i can find each.
Answers: 3


Chemistry, 22.06.2019 05:30, NorbxrtThaG
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1
Do you know the correct answer?
Questions in other subjects:

History, 14.10.2020 19:01



Mathematics, 14.10.2020 19:01

Mathematics, 14.10.2020 19:01



Social Studies, 14.10.2020 19:01

Mathematics, 14.10.2020 19:01

History, 14.10.2020 19:01