
Chemistry, 30.06.2021 02:10, alesiabarrios6
Na3N decomposes to form sodium and nitrogen gas at STP. If 13.7 L of nitrogen is produced
how many moles of Na3N was used? (22.4 L = 1 mole of any gas)
2Na3N --> 6Na + N2

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, alaina3792
Of the groups of elements below, which are most likely to gain electrons to become anions? a. alkali metal b. boron group c. halogen d. transition metal
Answers: 2


Chemistry, 22.06.2019 18:00, heggestade
To apply in a gold the individual gold atoms are united to each other by means of a metallic bond. how would you use the gold block to determine the atomic radius of a gold atom?
Answers: 3

Chemistry, 23.06.2019 01:00, aliviadushane
If a straight-chain hydrocarbon is a gas at room temperature, how many carbon atoms will it have? a. 6 carbon atoms b. 12 carbon atoms c. 24 carbon atoms d. 3 carbon atoms
Answers: 1
Do you know the correct answer?
Na3N decomposes to form sodium and nitrogen gas at STP. If 13.7 L of nitrogen is produced
how many...
Questions in other subjects:

Mathematics, 30.06.2019 16:00


Mathematics, 30.06.2019 16:00





Advanced Placement (AP), 30.06.2019 16:00

Mathematics, 30.06.2019 16:00

Mathematics, 30.06.2019 16:00


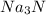 were used.
were used.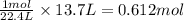
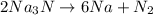
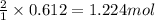 of
of 



