
Chemistry, 25.06.2021 05:40, genyjoannerubiera
For each system listed in the first column of the table below, decide (if possible) whether the change described in the second column will increase the entropy S of the system, decrease S, or leave S unchanged. If you don’t have enough information to decide, check the “not enough information “button in the last column.
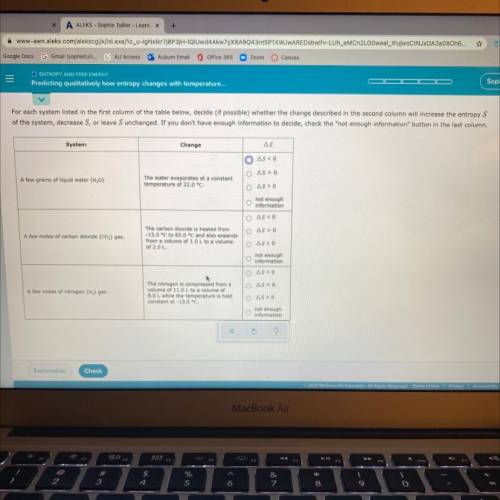

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, kingteron5870
Ionic compounds are made of ions, and yet the overall charge of an ionic compound is neutral. why?
Answers: 1


Chemistry, 22.06.2019 02:50, giiffnlojd
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 07:10, angellong94
Provide a stepwise curved arrow mechanism that fully explains the outcome of the reaction shown below. oh нао* heat он
Answers: 2
Do you know the correct answer?
For each system listed in the first column of the table below, decide (if possible) whether the chan...
Questions in other subjects:



Mathematics, 24.11.2020 18:20

Biology, 24.11.2020 18:20


Biology, 24.11.2020 18:20



Arts, 24.11.2020 18:20

Biology, 24.11.2020 18:20






