
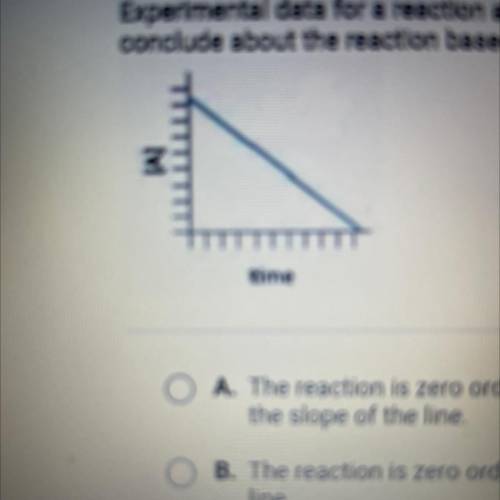
Experimental data for a reaction are collected and graphed. What can you
conclude about the reaction based on the graph?
A. The reaction is zero order, and the rate constant is the negative of
the slope of the line
B. The reaction is zero order, and the rate constant is the slope of the
line
C. The reaction is first order, and the rate constant is the slope of the
line,
D. The reaction is first order, and the rate constant is the negative eg

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, Bradgarner772
18. use the activity series to predict whether the following synthesis reaction will occur. write the chemical equations for the reaction if it's predicted to occur. (s) + o2(g) -> *note: it is possible.*
Answers: 1


Chemistry, 23.06.2019 00:30, emilylizbeth12334
Which of the following best describes technology a. something created for only scientists to use b. the method of thinking that scientists use. c. the application of engineering to create useful products. c. a scientific idea
Answers: 1

Chemistry, 23.06.2019 01:30, nikonee
Astudent states that 9.0 g of baking soda will form an unsaturated solution in 100 g of water. what do you need to know to decide whether this statement is correct? a. the temperature of the water and the molar mass of baking soda b. the percent by volume of the solution and the solubility of baking soda c. the temperature of the water and the solubility of baking soda at that temperature
Answers: 1
Do you know the correct answer?
Experimental data for a reaction are collected and graphed. What can you
conclude about the reactio...
Questions in other subjects:










History, 10.03.2020 17:00






