
Consider the following intermediate chemical equations.
CH2(g) →C(s) + 2H2(g)
CC1.(g) → C(s) +2012(g)
H2(g) +C12(g) → 2HCl(9)
ΔH, = 74.6 kJ
ΔH2 = 95.7 kJ
ΔΗ =
ΔHg =-92.3 kJ
What is the enthalpy of the overall chemical reaction CH (g) +4012(g) → CCl4(h)+ 4HCl(g)?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:50, hadwell34
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 15:30, lovebaeforlife351
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins. co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Do you know the correct answer?
Consider the following intermediate chemical equations.
CH2(g) →C(s) + 2H2(g)
CC1.(g) → C(s)...
CC1.(g) → C(s)...
Questions in other subjects:

Mathematics, 06.05.2021 21:30

Mathematics, 06.05.2021 21:30

Biology, 06.05.2021 21:30


Mathematics, 06.05.2021 21:30



Mathematics, 06.05.2021 21:30

History, 06.05.2021 21:30


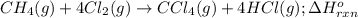
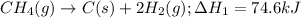
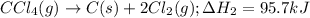
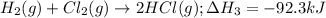
![\Delta H^o_{rxn}=[1\times (\Delta H_1)] + [1\times (-\Delta H_2)] + [2\times (\Delta H_3)]](/tpl/images/1382/1138/89421.png)
![\Delta H^o_{rxn}=[1\times (74.6)] + [1\times (-95.7)] + [2\times (-92.3)]\\\\\Delta H^o_{rxn}=-205.7kJ](/tpl/images/1382/1138/33de2.png)





