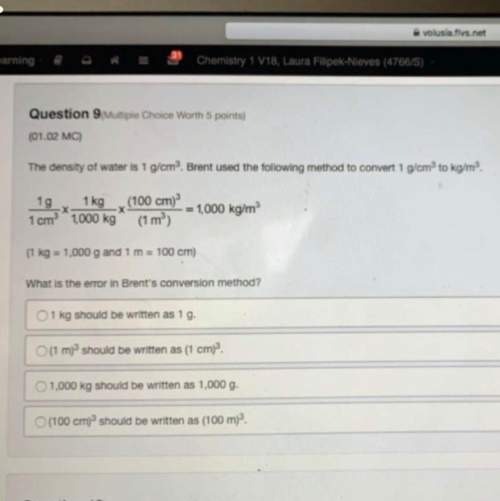
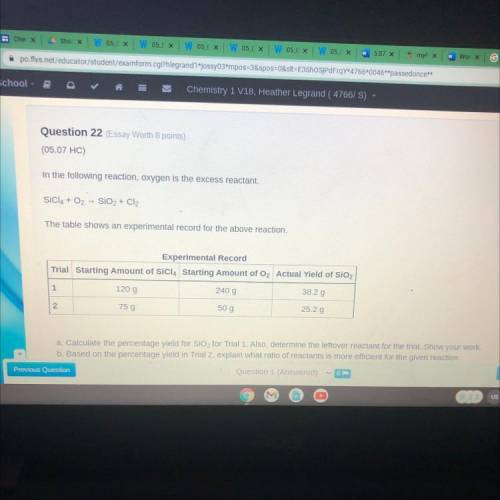
Question 22 (Essay Worth 8 points)
(05.07 HC)
In the following reaction, oxygen is the excess...

Question 22 (Essay Worth 8 points)
(05.07 HC)
In the following reaction, oxygen is the excess reactant.
SÍCIA + O2 - SiO2 + Cl2
The table shows an experimental record for the above reaction.
Experimental Record
Trial Starting Amount of SiCl4 Starting Amount of O2 Actual Yield of SiO2
1
120 g
240 g
38.2 g
N
75 g
50 g
25.2 g
a. Calculate the percentage yield for SiO2 for Trial 1. Also, determine the leftover reactant for the trial. Show your work
b. Based on the percentage yield in Trial 2, explain what ratio of reactants is more efficient for the given reaction.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 12:30, kitt90335
Asample contains 16.75 g of the radioisotope u-236 and 50.25 g of its daughter isotope, th-232. how long did it take for decay to take place if one half-life of u-236 is 23 million years? 46 million years 69 million years 92 million years 115 million years
Answers: 3

Chemistry, 21.06.2019 13:00, froyg1234
Read the given expression. x = number of protons − number of core electrons which of the following explains the identity of x and its trends across a period? x is the effective nuclear charge, and it remains constant across a period. x is the screening constant, and it remains constant across a period. x is the effective nuclear charge, and it increases across a period. x is the screening constant, and it increases across a period.
Answers: 1

Chemistry, 21.06.2019 16:00, WhiteWinterRose
What is the chemical formula of the following compound
Answers: 3

Do you know the correct answer?
Questions in other subjects:


History, 11.01.2021 18:50

Mathematics, 11.01.2021 18:50



Mathematics, 11.01.2021 18:50


Mathematics, 11.01.2021 18:50

English, 11.01.2021 18:50

Mathematics, 11.01.2021 18:50