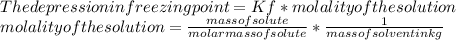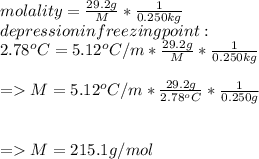Chemistry, 23.06.2021 02:50, jeffmacdonald1976
Suppose that you add 29.2 g of an unknown molecular compound to 0.250 kg of benzene, which has a K f of 5.12 oC/m. With the added solute, you find that there is a freezing point depression of 2.78 oC compared to pure benzene. What is the molar mass (in g/mol) of the unknown compound

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, brookekolmetz
How many orbitals does the p sub shell container
Answers: 3

Chemistry, 22.06.2019 00:30, TMeansStupidity
Jessica is traveling from miami, florida, to chicago, illinois. using the map, tell one way the land will change during the second half of her trip.
Answers: 1

Chemistry, 22.06.2019 04:00, amandasantiago2001
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3
Do you know the correct answer?
Suppose that you add 29.2 g of an unknown molecular compound to 0.250 kg of benzene, which has a K f...
Questions in other subjects:




History, 17.11.2020 20:40

Mathematics, 17.11.2020 20:40

Mathematics, 17.11.2020 20:40


Chemistry, 17.11.2020 20:40


Mathematics, 17.11.2020 20:40