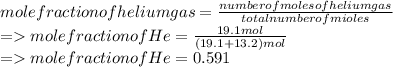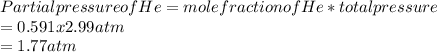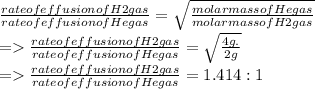Chemistry, 22.06.2021 06:00, pinkycupcakes3oxbqhx
A mixture of gases at 2.99 atm can You have two gases, and , at the same temperature. Determine the ratio of effusion rates of and .ists of 13.2 moles of hydrogen gas and 19.1 moles of helium gas. Determine the partial pressure of the helium gas.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:00, sammiehammer
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change. when the temperature in a room increases from 25°c to 33°c, changes from a solid to a liquid. in a lab, methane and nitrogen are cooled from -170°c to -200°c. the methane freezes and the nitrogen . when gold is heated to 2,856°c it changes from a liquid to a .
Answers: 2

Chemistry, 22.06.2019 11:30, ansuaprajita1506
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3

Chemistry, 23.06.2019 00:30, terryg4397
Fred is studying a substance that is made out of only one element. this means that
Answers: 1

Chemistry, 23.06.2019 03:00, duplessistoccara
Abaker touches a pie right after taking it out of the oven. which statement best explains why the pie feels hot?
Answers: 2
Do you know the correct answer?
A mixture of gases at 2.99 atm can You have two gases, and , at the same temperature. Determine the...
Questions in other subjects:

Mathematics, 21.06.2019 21:10


Mathematics, 21.06.2019 21:10


Biology, 21.06.2019 21:10