Chemistry, 21.06.2021 14:00, atsuedem974
Zinc sulfate is a 2-ion electrolyte,
dissociating 40% in a
certain concentration. Calculate its
dissociation (i) factor.
On the basis of 40% dissociation, 100
particles of zinc sulfate
will yield:
40zinc ions
40 sulfate ions
60undissociated particles
Jo 11:03

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 21:30, leenzazou587
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 23.06.2019 03:00, yoongislaugh
What volume does 1.70 ×10–3 mol of chlorine gas occupy if its temperature is 20.2 °c and its pressure is 795 mm hg?
Answers: 3
Do you know the correct answer?
Zinc sulfate is a 2-ion electrolyte,
dissociating 40% in a
certain concentration. Calculate i...
certain concentration. Calculate i...
Questions in other subjects:

Mathematics, 27.06.2020 19:01

Mathematics, 27.06.2020 19:01

Social Studies, 27.06.2020 19:01



English, 27.06.2020 19:01

Mathematics, 27.06.2020 19:01

Mathematics, 27.06.2020 19:01

History, 27.06.2020 19:01


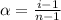
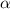 = degree of dissociation = 40% = 0.40
= degree of dissociation = 40% = 0.40