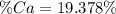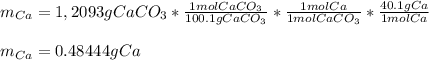Chemistry, 20.06.2021 17:40, jrfranckowiak
Una muestra de 2,5000 (g) de piedra caliza, se disuelve y se precipita el Calcio como oxalato de Calcio (CaC2O4), este precipitado se calcina transformándose en Carbonato de Calcio (CaCO3). El peso de este compuesto es de 1,2093 (g). Calcular el % de Calcio en la muestra

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, jzjajsbdb8035
Which other elements contain the same number of outer electrons as sodium
Answers: 3

Chemistry, 22.06.2019 15:00, kamkam5791
Is powdered sports drink ionic or covalent ? 10pts !
Answers: 1

Chemistry, 22.06.2019 18:00, brisacruz013
Which statement best describes the he properties of iconic compounds ?
Answers: 1
Do you know the correct answer?
Una muestra de 2,5000 (g) de piedra caliza, se disuelve y se precipita el Calcio como oxalato de Cal...
Questions in other subjects:

Mathematics, 16.04.2020 21:28

Social Studies, 16.04.2020 21:28

Mathematics, 16.04.2020 21:28

History, 16.04.2020 21:28


Mathematics, 16.04.2020 21:28