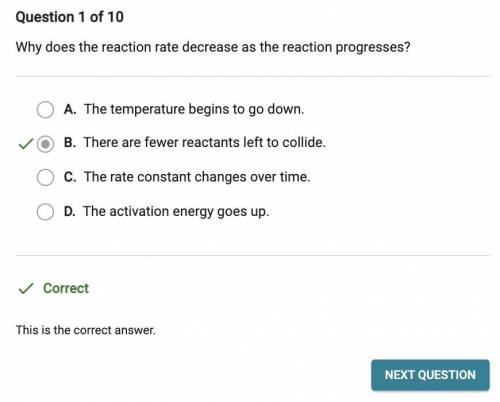
Chemistry, 17.06.2021 21:50, clairebear65
Why does the reaction rate decrease as the reaction progresses?
O A. The temperature begins to go down.
B. The activation energy goes up.
O C. The rate constant changes over time.
O D. There are fewer reactants left to collide.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:00, tchase0616
What is the molarity of 60.0 grams of naoh dissolved in 750 milliliters of water? a) 1.1 m b) 2.0 m c) 12 m d) 75 m
Answers: 1

Chemistry, 22.06.2019 08:30, masontdavis
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 16:50, TrueKing184
Answer asap need by wednesday morning explain how a buffer works, using an ethanoic acid/sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 1
Do you know the correct answer?
Why does the reaction rate decrease as the reaction progresses?
O A. The temperature begins to go d...
Questions in other subjects:


History, 04.07.2019 11:00


Computers and Technology, 04.07.2019 11:00


Biology, 04.07.2019 11:00


Biology, 04.07.2019 11:00