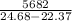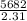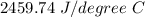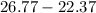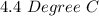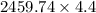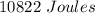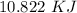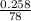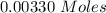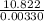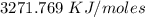Chemistry, 16.06.2021 23:10, ejcastilllo
Two experiments were conducted in a bomb calorimeter. The first one to determine the heat capacity of the calorimeter, the second the heat of combustion of the carcinogenic substance benzene (C6H6). a. In the first experiment, the temperature rises from 22.37 o C to 24.68 o C when the calorimeter absorbs 5682 J of heat. Determine the heat capacity of the calorimeter. Page 3 of 4 b. In the second experiment, the combustion of 0.258 g of benzene increases the temperature from 22.37 o C to 26.77 o C. Determine the heat of combustion for 1 mol of benzene.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:10, cheesedoodle
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1
Do you know the correct answer?
Two experiments were conducted in a bomb calorimeter. The first one to determine the heat capacity o...
Questions in other subjects:


Social Studies, 02.12.2021 03:00


Business, 02.12.2021 03:00

Social Studies, 02.12.2021 03:00

Chemistry, 02.12.2021 03:00

History, 02.12.2021 03:00

Computers and Technology, 02.12.2021 03:00