What amount of oxygen gas is needed to produce 2.85 mol of chlorine gas……
NEED HELP ASAP!
A)...

Chemistry, 16.06.2021 18:30, jahnoibenjamin
What amount of oxygen gas is needed to produce 2.85 mol of chlorine gas……
NEED HELP ASAP!
A) 0.95 mol
B) 1.90 mol
C) 2.85 mol
D) 5.70 mol
E) 8.55 mol
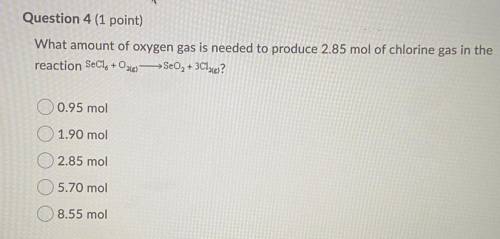

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, microwave13016
Agroup of students is studying convection current. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other is in an area with warm air. after 10 minutes, the balloon are released from a height of 1 meter. which of the following to the students most likely observe? a) the warm balloon expands and rises. the cold balloon shrinks and sinks b) the balloon both rise. the cold balloon is larger than the warm balloon c) the cold balloon expands and rises. the warm balloon shrinks and sinks d) the balloon rise at the same rate. both balloons are the same size
Answers: 1


Chemistry, 22.06.2019 18:10, sangamlama
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 22.06.2019 22:20, trockout4868
How do cfcs cause ozone depletion? how do cfcs cause ozone depletion? ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule.
Answers: 2
Do you know the correct answer?
Questions in other subjects:

Engineering, 09.01.2022 16:50




Mathematics, 09.01.2022 17:00


English, 09.01.2022 17:00

Mathematics, 09.01.2022 17:00


Advanced Placement (AP), 09.01.2022 17:00






