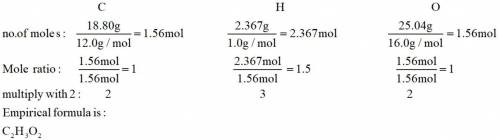Chemistry, 16.06.2021 04:00, mathiscool51
Analysis of a sample of a compound composed of carbon, hydrogen, and oxygen shows that the sample contains 18.80 g of C, 2.367 g of H, and 25.04 g of O. The properties of the compound suggest that the molar mass should be 59.04 g/mol. How many carbon atoms are there in one molecule of the compound

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, Cythina2007
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1


Chemistry, 22.06.2019 22:30, eduardoguizar8787
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1

Chemistry, 23.06.2019 04:00, hailey200127
What is the volume of 2.5 moles of nitrogen gas (n2)at standard temperature and pressure (stp)?
Answers: 1
Do you know the correct answer?
Analysis of a sample of a compound composed of carbon, hydrogen, and oxygen shows that the sample co...
Questions in other subjects:



Mathematics, 04.09.2019 00:30



Biology, 04.09.2019 00:30

Mathematics, 04.09.2019 00:30


Mathematics, 04.09.2019 00:30


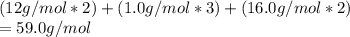
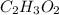 .
.