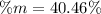Chemistry, 16.06.2021 01:00, chodister353
A mixture of BaCl2 and NaCl is analyzed by precipitating all the barium as BaSO4. After addition of an excess of Na2SO4 to a 3.656-g sample of the mixture, the mass of precipitate collected is 1.658 g. What is the mass percentage of barium chloride in the mixture

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:00, MilanPatel
How is the shape of the poem “peer” connected to its meaning?
Answers: 2

Chemistry, 23.06.2019 02:00, hannabeth91
When an experimenter draws a conclusion that he assumes will apply to all situations set up similarly to his test situation, even though he cannot possibly have examined all possible test scenarios, the experimenter is using deductive reasoning inductive reasoning abductive reasoning subjective reasoning
Answers: 1

Chemistry, 23.06.2019 06:30, SimplyGenesis762
Generally, observed behavior that can be formulated into a statement, sometimes mathematical in nature, is called a(n): a. observation. b. measurement. c. theory. d. natural law. e. experiment.
Answers: 2

Chemistry, 23.06.2019 09:30, Dariana123
Which of the following is not a characteristic of a hydrogen bond? 1. it is responsible for the unusual physical properties of water. 2. it is weaker than a covalent bond. 3. it is stronger than other dipole-dipole interactions. 4. it can occur when hydrogen is covalently bound to very electronegative elements liks f, cl, br and i.
Answers: 1
Do you know the correct answer?
A mixture of BaCl2 and NaCl is analyzed by precipitating all the barium as BaSO4. After addition of...
Questions in other subjects:



History, 30.06.2019 08:00

Mathematics, 30.06.2019 08:00

Mathematics, 30.06.2019 08:00

Biology, 30.06.2019 08:00

Mathematics, 30.06.2019 08:00

Mathematics, 30.06.2019 08:00

Mathematics, 30.06.2019 08:00

Mathematics, 30.06.2019 08:00