
Chemistry, 14.06.2021 20:10, eduardoma2902
Find the boiling point of a solution of 2.00 m solution of sodium chloride, NaCl, in water (kb= 0.512°C, bp= 100.0°C)

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 05:00, cpcoolestkid4
C=59(f−32)the equation above shows how temperature f, measured in degrees fahrenheit, relates to a temperature c, measured in degrees celsius. based on the equation, which of the following must be true? a temperature increase of 1 degree fahrenheit is equivalent to a temperature increase of 59 degree celsius. a temperature increase of 1 degree celsius is equivalent to a temperature increase of 1.8 degrees fahrenheit. a temperature increase of 59 degree fahrenheit is equivalent to a temperature increase of 1 degree celsius. a) i onlyb) ii onlyc) iii onlyd) i and ii only
Answers: 1

Chemistry, 23.06.2019 07:00, asims13
The following transition occurs at a molecular level for a substance. what transition corresponds to this change in microscopic structure? the carbon dioxide molecules on the left are in a regular, tightly packed pattern. after heating, it becomes much lower density. a. melting b. boiling c. sublimation d. freezing
Answers: 1

Chemistry, 23.06.2019 18:00, hellokitty1647
Based on the data in the map where would you expect vegitation on earth to be more dense
Answers: 2
Do you know the correct answer?
Find the boiling point of a solution of 2.00 m solution of sodium chloride, NaCl, in water (kb= 0.51...
Questions in other subjects:

Mathematics, 22.02.2021 17:10

Mathematics, 22.02.2021 17:10





History, 22.02.2021 17:10

Mathematics, 22.02.2021 17:10



 .
.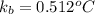
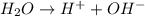
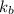 = molal boiling point elevation constant
= molal boiling point elevation constant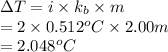
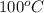 . Hence, the boiling point of solution will be as follows.
. Hence, the boiling point of solution will be as follows.





