Chemistry, 11.06.2021 06:50, smcardenas02
What is the delta H when 72.0 grams H2O condenses at 100.00C?
Here are some constants that you MAY need.
specific heats heat of fusion heat of vaporization
H2O(s) = 2.1 J/g0C 6.01 kJ/mole 40.7 kJ/mole
H2O(L) = 4.18 J/g0C
H2O(g) = 1.7 J/g0C
2930 kJ
163 kJ
-163 kJ
-2930 kJ

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 22:30, darceline1574
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Do you know the correct answer?
What is the delta H when 72.0 grams H2O condenses at 100.00C?
Here are some constants that you MAY...
Questions in other subjects:





Mathematics, 14.07.2019 05:10

Mathematics, 14.07.2019 05:10





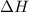 is -163 kJZ
is -163 kJZ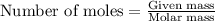 ......(1)
......(1)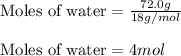
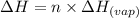 ......(2)
......(2)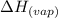 = specific heat of vaporization = -40.7 kJ/mol
= specific heat of vaporization = -40.7 kJ/mol