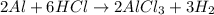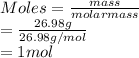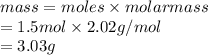Chemistry, 10.06.2021 15:20, drandbone92
Calculate the mass of hydrogen formed when 26.98 g of aluminum reacts with excess hydrochloric acid according to the following balanced chemical equation: 2 Al + 6 HCl → 2 AlCl3 + 3 H2

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, ashleyjaslin
Calculate the expected ph values of the buffer systems from the experiments (a, b,c, d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2


Chemistry, 22.06.2019 07:30, isalih7256
Free answer. the treaty of versailles ended world war i, but some of the terms of the treaty contributed to the beginning of world war ii. which was one of the terms of the treaty? the answer would be "germany was forces to pay reparations to the allied countries.". i hope this .
Answers: 1
Do you know the correct answer?
Calculate the mass of hydrogen formed when 26.98 g of aluminum reacts with excess hydrochloric acid...
Questions in other subjects:

Social Studies, 21.09.2019 03:30


History, 21.09.2019 03:30



History, 21.09.2019 03:30

Biology, 21.09.2019 03:30

Advanced Placement (AP), 21.09.2019 03:30


Biology, 21.09.2019 03:30