
Chemistry, 09.06.2021 09:50, aliceohern
A solution containing 1.22 g of a diprotic acid H2CH2O4 (malonic acid)
was titrated with 45.5 mL of NaOH to reach the second equivalence
point. What is the concentration of the NaOH solution? (MW_malonic
acid = 104.06 g/mol)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, mrylenastewart
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1



Chemistry, 23.06.2019 00:50, alainacorkell6472
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1
Do you know the correct answer?
A solution containing 1.22 g of a diprotic acid H2CH2O4 (malonic acid)
was titrated with 45.5 mL of...
Questions in other subjects:

Biology, 26.05.2020 23:02

Biology, 26.05.2020 23:02

Mathematics, 26.05.2020 23:02



Spanish, 26.05.2020 23:02

Mathematics, 26.05.2020 23:02

Mathematics, 26.05.2020 23:02



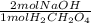 = 0.02344 mol NaOH
= 0.02344 mol NaOH



