
For the equilibrium that exists in an aqueous solution of nitrous acid (HNO2, a weak acid), the equilibrium constant expression is:
a. K = [ H+] [NO2-] / [HNO2]
b. K = [ H+] [N] [O]2 / [HNO2]
c. K = [ H+] [NO2-] / [HNO2]
d. K = [H+]2 [NO2-] / [HNO2]
e. None of these

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, 20alondra04
In order to calculate the amount of heat transferred you must know the __ and specific heat of the material, as well as the change in temperature. a. volume b. density c. mass d. enthalpy
Answers: 1

Chemistry, 22.06.2019 10:00, ellaemtagedeane
Nonpoint source pollution is difficult to control because it
Answers: 2

Chemistry, 22.06.2019 11:40, tatemelliott
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 12:00, BakerElsie02
Which of the following units is not an official si unit? mole liter kilogram ampere
Answers: 1
Do you know the correct answer?
For the equilibrium that exists in an aqueous solution of nitrous acid (HNO2, a weak acid), the equi...
Questions in other subjects:




Health, 29.09.2019 05:10




Social Studies, 29.09.2019 05:10

Arts, 29.09.2019 05:10


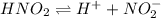
![K = \frac{[H^{+}][NO^{-}_{2}]}{[HNO_{2}]}](/tpl/images/1365/7220/b80db.png)




