
Chemistry, 05.06.2021 22:50, Kaysofine11icloudcom
What is the limiting reactant in a reaction where 10.0 mol of iron is treated with 12.0 mol of bromine? The product that forms is FeBr3. First, write and balance the chemical equation. Next, calculate the moles of FeBr3 that can be made from 10.0 mol of Fe. Then calculate the moles of FeBr3 that can be made from 12.0 mol of bromine. The smaller amount of FeBr3 reveals the limiting reactant.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:40, trinityanne1738
Asolid that forms and separates from a liquid mixture is called
Answers: 2


Chemistry, 23.06.2019 01:00, jaidencoolman2510
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2
Do you know the correct answer?
What is the limiting reactant in a reaction where 10.0 mol of iron is treated with 12.0 mol of bromi...
Questions in other subjects:



Mathematics, 23.04.2020 22:25

English, 23.04.2020 22:25

Chemistry, 23.04.2020 22:25




Biology, 23.04.2020 22:25


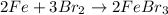
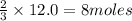 of iron
of iron



