
Chemistry, 04.06.2021 04:10, jazmaine1217
PLEASE HELP ME! I really can’t fail chemistry I tried to do it myself but I feel like my brain doesn’t function properly with stuff like this
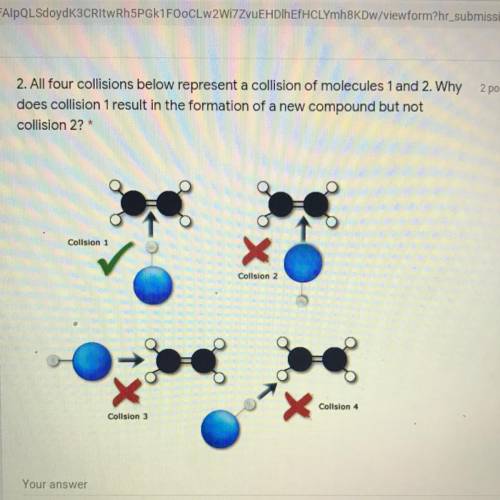

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, codeyhatch142
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1


Do you know the correct answer?
PLEASE HELP ME! I really can’t fail chemistry I tried to do it myself but I feel like my brain doesn...
Questions in other subjects:


World Languages, 25.09.2020 14:01




Mathematics, 25.09.2020 14:01



Mathematics, 25.09.2020 14:01

History, 25.09.2020 14:01






