Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:30, nekathadon
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1


Chemistry, 22.06.2019 18:10, sangamlama
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1
Do you know the correct answer?
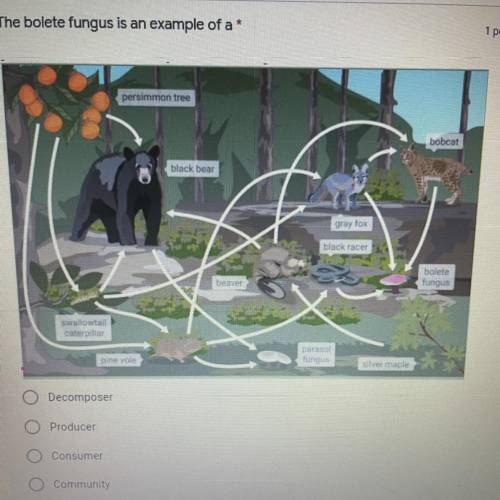
The bolete fungus is an example of a
...
...
Questions in other subjects:

History, 09.07.2019 23:00

Physics, 09.07.2019 23:00


Mathematics, 09.07.2019 23:00

Mathematics, 09.07.2019 23:00


Mathematics, 09.07.2019 23:00

Health, 09.07.2019 23:00