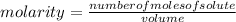Chemistry, 03.06.2021 07:20, yselahernandez02
a chemist dissolves 0.564 moles of manganese (IV) oxide (MnO2) in water, and adds enough water to make 0.510 L of solution. Calculate the molarity of the solution.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, jonloya264
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1



Chemistry, 22.06.2019 20:10, sarahalexa19
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2
Do you know the correct answer?
a chemist dissolves 0.564 moles of manganese (IV) oxide (MnO2) in water, and adds enough water to ma...
Questions in other subjects:



Spanish, 09.04.2021 20:00

Mathematics, 09.04.2021 20:00

Mathematics, 09.04.2021 20:00

Mathematics, 09.04.2021 20:00




Mathematics, 09.04.2021 20:00