
An investigation is conducted into how the mass of magnesium metal reacting with hydrochloric acid affects the amount of hydrogen gas produced.
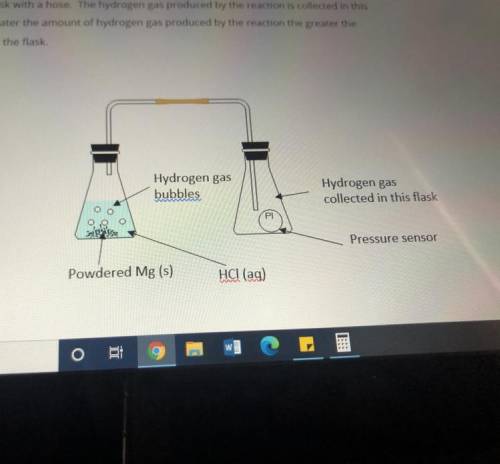
Masses of 0.10g, 0.20g, 0.30g and 0.40g of powdered Mg metal are reacted with hydrochloric acid(HCl). The conical flask containing the reaction mixture of Mg and HCl is connected to another conical flask with a hose. The hydrogen gas produced by the reaction is collected in this conical flask. The greater the amount of hydrogen gas produced by the reaction the greater the pressure of the gas in the flask.
A) what is the independent variable:
B) what is the dependent variable:
C) write a hypothesis for this investigation:
D) give 2 variables that should have been controlled for this investigation:
thank you sm!!!

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 16:50, shaylawaldo11
Which element is least likely to undergo a chemical reaction
Answers: 3

Do you know the correct answer?
An investigation is conducted into how the mass of magnesium metal reacting with hydrochloric acid a...
Questions in other subjects:



History, 31.03.2020 21:43


Mathematics, 31.03.2020 21:43


Chemistry, 31.03.2020 21:43

Geography, 31.03.2020 21:43

English, 31.03.2020 21:44

English, 31.03.2020 21:44






