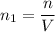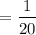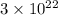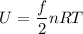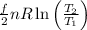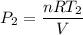A quarter of a mole of neon gas is in equilibrium at 330 K in a container that has a volume of 5.0L.
(a) How many atoms of neon would you expect to find in one portion of the container that has a volume of 1.0L? Explain your answer in terms of the definition of equilibrium given in our study of entropy.
(b) What is the thermal energy of the gas in this state?
(c) If the container is heated so that the gas reaches a temperature of 380 K, what is the change in the entropy of the system? Assume that the expansion of the container is negligible. (If you can give a number, do so; otherwise, at least say whether the entropy increases or decreases.)
(d) If the expansion of the container is negligible, what must the pressure of the heated gas be?

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 09:30, kevinh2683
Apump contains 0.5 l of air at 203 kpa. you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2

Chemistry, 22.06.2019 15:30, vivianfling
Why does earth rotate? because earth is formed from cold gases collapsing due to gravity because the matter in the nebula that formed earth was spinning because earth forms more than 99% of the mass of the solar system because the hydrogen atoms inside the nebula fused to form helium
Answers: 1
Do you know the correct answer?
A quarter of a mole of neon gas is in equilibrium at 330 K in a container that has a volume of 5.0L....
Questions in other subjects:

Mathematics, 08.09.2020 01:01







Mathematics, 08.09.2020 01:01

Mathematics, 08.09.2020 01:01