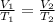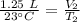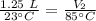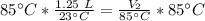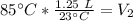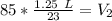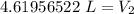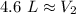Chemistry, 27.05.2021 06:40, carolinamleal04
A balloon containing helium gas has a volume of 1.25 L at room temperature (23 oC). The balloon is heated to at temperature of 85 oC . Assuming no change in pressure, what is the new volume of the balloon?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, bibhu42kumarp7o4ss
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1



Chemistry, 23.06.2019 01:00, jaidencoolman2510
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2
Do you know the correct answer?
A balloon containing helium gas has a volume of 1.25 L at room temperature (23 oC). The balloon is h...
Questions in other subjects:

History, 29.10.2019 17:31

Physics, 29.10.2019 17:31


English, 29.10.2019 17:31


Medicine, 29.10.2019 17:31

English, 29.10.2019 17:31


Mathematics, 29.10.2019 17:31

English, 29.10.2019 17:31