
Chemistry, 27.05.2021 05:20, wrightlilybug07
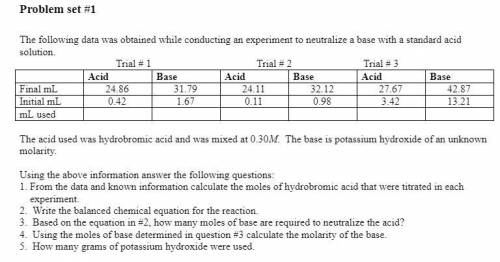
Using the above information answer the following questions:
1. From the data and known information calculate the moles of hydrobromic acid that were titrated in each
experiment.
2. Write the balanced chemical equation for the reaction.
3. Based on the equation in #2, how many moles of base are required to neutralize the acid?
4. Using the moles of base determined in question #3 calculate the molarity of the base.
5. How many grams of potassium hydroxide were used.

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 01:50, lildestinyquintana
Ase your answer to this question on the information below. hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant. the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen. chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product. nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1


Chemistry, 22.06.2019 12:00, luffybunny
There is one girl i like and i don't know how to tell her that, i have a feeling she knows but if she doesn't i don't want to make a fool out of myself how is one way to boost my confidence on asking her out
Answers: 1
Do you know the correct answer?
Using the above information answer the following questions:
1. From the data and known information...
Questions in other subjects:


Social Studies, 19.02.2021 22:50



Chemistry, 19.02.2021 22:50

Mathematics, 19.02.2021 22:50

Mathematics, 19.02.2021 22:50

Mathematics, 19.02.2021 22:50

Mathematics, 19.02.2021 22:50






