
Chemistry, 26.05.2021 01:00, emilystartk
) Dinitrogen Tetroxide partially decomposes according to the following equilibrium: N2O4 (g) 2NO2 (g) A 1.00-L flask is charged with 0.400 mol of N2O4. At equilibrium at 373 K, 0.0055 mol of N2O4 remains. Keq for this reaction is .

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 06:40, CylieTbh
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3

Chemistry, 22.06.2019 09:00, stelllllllllllllllla
George is a dalmatian puppy. describe what happens to light that allows you to see george’s black and white coat.
Answers: 1
Do you know the correct answer?
) Dinitrogen Tetroxide partially decomposes according to the following equilibrium: N2O4 (g) 2NO2 (g...
Questions in other subjects:

English, 24.09.2021 01:10

Engineering, 24.09.2021 01:10


Mathematics, 24.09.2021 01:10

World Languages, 24.09.2021 01:10




English, 24.09.2021 01:10


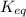 for this reaction is 1.578.
for this reaction is 1.578.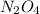 = 0.4 mol
= 0.4 mol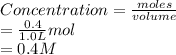
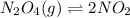
![[NO_{2}]](/tpl/images/1347/8951/53e25.png) is calculated as follows.
is calculated as follows.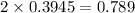
![K_{eq} = \frac{[NO_{2}]^{2}}{[N_{2}O_{4}]}\\= \frac{(0.789)^{2}}{(0.3945)}\\= 1.578](/tpl/images/1347/8951/12c3a.png)




