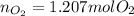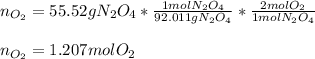1N2 + 2O2 → 1N2O4
If 55.52 grams of N2O4 was produced, how many moles of O2 were requir...

Chemistry, 21.05.2021 06:50, SunsetPrincess
1N2 + 2O2 → 1N2O4
If 55.52 grams of N2O4 was produced, how many moles of O2 were required?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 00:00, lilyclairehutson
Which of the following statements is true? a. elements in the last period are radioactive. b. atomic weight is the same as atomic mass. c. elements in the same group have the same number of electron shells. d. atomic number equals the number of neutrons in the nucleus of an atom.
Answers: 1

Chemistry, 22.06.2019 12:00, Unknowndragon42
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2
Do you know the correct answer?
Questions in other subjects: