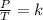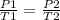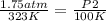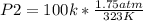Chemistry, 21.05.2021 03:40, gnarlyjordan
A gas has a volume of 300 mL in a rigid container at 50oC and 1.75 atm. What will be its pressure at 100K?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:00, cathydaves
What is the chemical formula of the following compound
Answers: 1

Chemistry, 21.06.2019 20:30, pennygillbert
In which layer of earth do most earthauakes occur a_ inner core b_outer core c_mantle d_crust
Answers: 1

Chemistry, 22.06.2019 10:40, yfgkeyonna
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2
Do you know the correct answer?
A gas has a volume of 300 mL in a rigid container at 50oC and 1.75 atm. What will be its pressure at...
Questions in other subjects:



Health, 06.11.2020 15:30




Mathematics, 06.11.2020 15:30


English, 06.11.2020 15:30