
Chemistry, 20.05.2021 02:50, squidywar297
COULD SOMEONE PLEASE GIVE ME THE ANSWER FOR THIS NO BOTS PLEASEEE TODAY IS THE LAST DAY FOR MISSING WORK
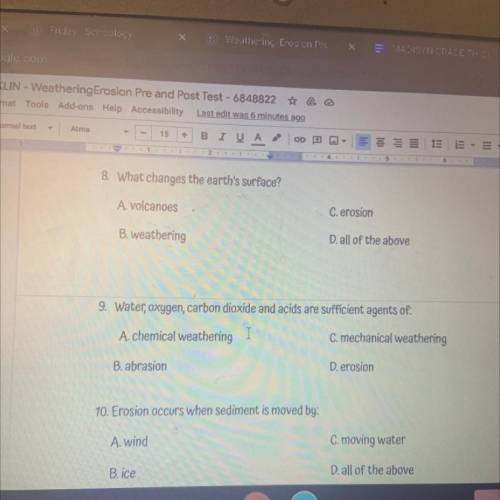

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, gwenparks
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 12:30, azzyla2003
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

Do you know the correct answer?
COULD SOMEONE PLEASE GIVE ME THE ANSWER FOR THIS NO BOTS PLEASEEE TODAY IS THE LAST DAY FOR MISSING...
Questions in other subjects:


History, 05.05.2021 23:30


Mathematics, 05.05.2021 23:30


Computers and Technology, 05.05.2021 23:30

Mathematics, 05.05.2021 23:30



Chemistry, 05.05.2021 23:30






