
Chemistry, 18.05.2021 21:10, marahsenno
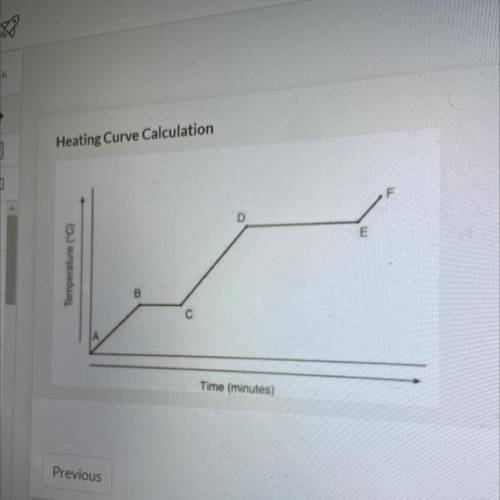
Referencing the figure to the left, how much energy in J would be
needed to change 59 g of ice at -22.75 °C into steam at 128.65 °C
Important numbers/equations:
q = Cpm: AT
Specific Heat of Water (liquid) = 4.18 J/go°C
Specific Heat of Water (solid) = 2.06 J/g•°C
Specific Heat of Water (gas) = 2.01 J/g °C
Molar Heat of Fusion of water: 6009 J/mol
Molar Heat of Vaporization for water: 40790 J/mol

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:50, britotellerialuis
Evaluate this exponential expression,8. (2 + 3)2 – 42
Answers: 3

Chemistry, 22.06.2019 06:30, Pizzapegasus1
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1


Chemistry, 22.06.2019 19:20, Lovelybunny321
15. which of the following is not human-caused groundwater pollution? a. water in an aquifer dissolves elements such as arsenic and mercury from surrounding rock. b. water in an aquifer is contaminated by leachate that seeps into the ground from a landfill. c. water in an aquifer becomes polluted with chemicals used in hydraulic fracturing, or fracking. d. water in an aquifer absorbs harmful bacteria from the drainage field of a septic tank.
Answers: 1
Do you know the correct answer?
Referencing the figure to the left, how much energy in J would be
needed to change 59 g of ice at -...
Questions in other subjects:

English, 08.04.2021 21:30



Mathematics, 08.04.2021 21:30

English, 08.04.2021 21:30


Mathematics, 08.04.2021 21:30

Mathematics, 08.04.2021 21:30

Mathematics, 08.04.2021 21:30






