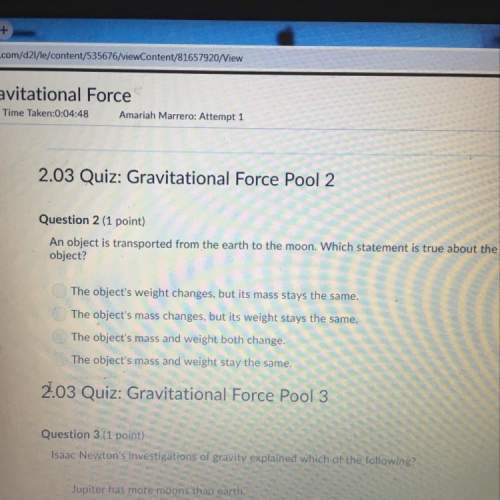
Chemistry, 17.05.2021 19:30, corvettapj6583
7. How much 0.5 M KCl solution is required to react completely with 100 mL of 2M AgNO3 solution?
KCl + AgNO3 → AgCl + KNO3
8. In the previous question, the AgCl produced is nonsoluble and forms a precipitate. If this precipitate is weighed and dried, what mass of AgCl would you expect to find?
9. A fixed volume of 15 cm3 of N2 at STP is reacted with excess H2. What volume of NH3 is produced at STP?
N2 + 3H2 → 2NH3
10. 50 mL of 2.0M NaBr is reacted with excess Cl2. The Br2 gas produced is collected. What volume would the gas occupy at 298 K and 1 atm?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:00, rafaelasoareschagas7
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3


Chemistry, 23.06.2019 00:50, maddysmall32
Which of the following warnings would an agricultural chemist tell a farmer who wants to recycle his or her own ammonia? recycling ammonia is a difficult process that sometimes takes weeks. recycling ammonia requires a degree in biochemistry or a related field. recycling ammonia can be harmful because it is highly flammable and toxic. recycling ammonia costs too much money considering the price of the necessary chemicals.
Answers: 1
Do you know the correct answer?
7. How much 0.5 M KCl solution is required to react completely with 100 mL of 2M AgNO3 solution?
KC...
Questions in other subjects:

Mathematics, 25.12.2021 23:50


Mathematics, 25.12.2021 23:50

Geography, 25.12.2021 23:50


Arts, 25.12.2021 23:50

Biology, 25.12.2021 23:50


Mathematics, 25.12.2021 23:50

Mathematics, 25.12.2021 23:50