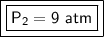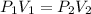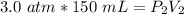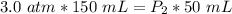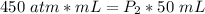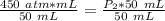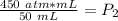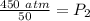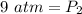Chemistry, 17.05.2021 03:50, jacobmclawhorn3001
What is the new pressure of 150 mL of a gas that is compressed to 50 mL when the original pressure was 3.0 ATM and the temperature is held constant

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:00, asanchez4292
What type of matter is made of only one kind of atom
Answers: 2

Chemistry, 22.06.2019 14:40, neonbluefaith
Which statement best describes the function of enzymes?
Answers: 1

Chemistry, 23.06.2019 07:30, jessicawelch25
In a laboratory determination of the atomic weight of tin, a sample of tin is weighed in a crucible. nitric acid is added, and the reaction proceeds to give a hydrated tin(iv)oxide plus no2and h2o. the hydrated tin(iv)oxide is then heated strongly and reacts as follows: sno2.xh2o(s)sno2(s)+ xh2o(g)the sno2is finally cooled and weighed in the crucible. explain the effect on the calculated atomic weight of tin that would result from each of the following experimental errors: (a)considerable spattering occurs when the nitric acid is added to the tin.(b)the hydrated tin(iv)oxide is not heated sufficiently to change it completely to tin oxide.
Answers: 2
Do you know the correct answer?
What is the new pressure of 150 mL of a gas that is compressed to 50 mL when the original pressure w...
Questions in other subjects:




English, 22.11.2019 18:31

Mathematics, 22.11.2019 18:31



History, 22.11.2019 18:31


History, 22.11.2019 18:31