
Chemistry, 16.05.2021 02:20, bunnybuge4
A large balloon contains 11.7 g of helium. What volume will the helium occupy at an altitude of 10 000 m, where the atmospheric pressure is 199.12 mmHg and the temperature is -50.0 C
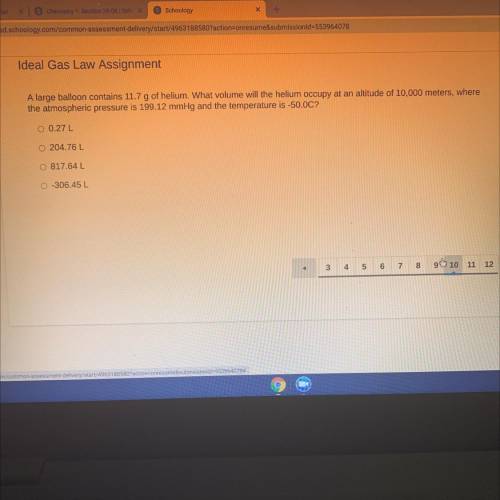

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:10, andybiersack154
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1



Chemistry, 22.06.2019 19:50, VoidedAngel
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3
Do you know the correct answer?
A large balloon contains 11.7 g of helium. What volume will the helium occupy at an altitude of 10 0...
Questions in other subjects:

Biology, 15.01.2021 23:20

Health, 15.01.2021 23:20

Mathematics, 15.01.2021 23:20


Spanish, 15.01.2021 23:20

Mathematics, 15.01.2021 23:20

Mathematics, 15.01.2021 23:20









